E-cigarettes: effects on cardiovascular system
| Take Home Messages |
|---|
|
Introduction
Tobacco use is the principal cause of preventable death worldwide.1 According to the World Health Organization (WHO), approximately 100 million people were killed in the 20th century as a result of tobacco consumption.1 Most of the danger associated with tobacco smoking stems from combustion, which creates more than 7000 chemicals, of which at least 70 are known to be carcinogenic.2 Approximately 28% of the 480 000 annual deaths attributed to cigarette smoking in the United States are caused by ischaemic heart disease.1
Electronic cigarette devices
Electronic cigarette devices (e-cigarettes) provide an alternative to conventional cigarette smoking that allow users to inhale nicotine in a vapour (see Figure 1).1,2 Equipped with a nicotine cartridge, vaporizing chamber, heating coil, atomizer and rechargeable battery,3 they typically work by heating a liquid that usually contains nicotine, propylene glycol or vegetable glycerine, with flavourings; and can take the form of cigalikes, vape pens, pod systems or mods with refillable tanks (see Table 1).4 As opposed to smoking, E-cigarettes do not burn tobacco and therefore do not produce tar or carbon monoxide, which are two of the most harmful by-products in tobacco smoke. E-cigarettes are currently unavailable on the National Health Service prescription.5
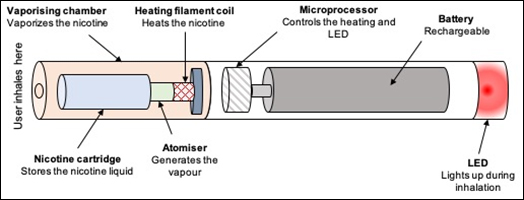
Figure 1. E-cigarette design (adapted from 12)
LED light emitting diode
Table 1. Types of e-cigarettes5 | ||
|---|---|---|
Type | How they work | |
Cigalike, rechargeable e-cigarette
|
| Look similar to tobacco cigarettes, have small batteries and can be disposable or rechargeable. |
Pod systems |
| Compact, rechargeable. Operating with pre-filled or refillable liquid. |
Mods |
| Largest e-cigarette device with longer lasting batteries and variable power. |
Vape pens, personal vaporiser |
| Shaped like a pen, they have a tank to store e-liquid and replaceable coils. The batteries tend to last longer than cigalike batteries and are rechargeable. |
a Licensed under the Creative Commons Attribution-Share Alike 3.0 Unported license Available at https://commons.wikimedia.org/wiki/File:Cig_a_like--_2013-10-21_03-47.jpg b This file is licensed under the Creative Commons Attribution-Share Alike 4.0 International license. Available at https://commons.wikimedia.org/wiki/File:Juul_vaping_device_with_pods_(cropped).jpg c This file is licensed under the Creative Commons Attribution-Share Alike 2.0 Generic license. https://commons.wikimedia.org/wiki/File:E_Cigarettes,_Ego,_Vaporizers_and_Box_Mods_(17679064871).jpg d This file is licensed under the Creative Commons Attribution-Share Alike 4.0 International license. Available at https://commons.wikimedia.org/wiki/File:Electronic_cigarette_vape_pen.JPG The images were modified (cropped). | ||
The use of e-cigarettes has been growing in recent years, with an estimated 3.6 million (7.1%) of the adult population in the UK presently using E-cigarettes. The peak age range of users in 2019 was 35-44 years.5 About half (54.1%) of current vapers are ex-smokers;6 the main reasons given for switching to e-cigarettes were to quit and to prevent relapse.6 Within the respiratory community there was controversy surrounding the utility of e-cigarettes; advocates emphasise they significantly reduce risk compared to tobacco cigarettes. Whilst others raised concerns surrounding their safety. Following an outbreak of vaping-induced injury (mechanisms include acute eosinophilic pneumonia, respiratory bronchiolitis-associated interstitial lung disease and hypersensitivity pneumonitis),8 the respiratory community now largely discourage the use of e-cigarettes. There however remains insufficient high-quality evidence on the short and long-term safety of these devices. Observational studies in the 2016 United States of America National Health Interview surveys reported that although the risk of myocardial infarction remains greater with tobacco cigarettes (odds ratio 2.72, 95% confidence interval (CI) 2.29-3.24), daily e-cigarette use was also associated with significantly higher risk of myocardial infarction (odds ratio 1.79, 95% CI 1.20-2.66).6,7
Cardiovascular effects of E-cigarettes
It has been advanced that the main cardiovascular side effects of e-cigarettes could be due to possible nicotine effects, as well as harmful components such as aldehydes (e.g. acrolein), and fine and ultrafine particles (PMs) (see Table 2 and Figure 2).12 There is currently very limited data on the vascular effects of e-cigarettes smoking. Some studies assessing acrolein inhalation from smoking and ambient air pollution have previously reported deleterious effects of acrolein: increasing vascular injury by impairing vascular repair capacity; increasing the risk of thrombosis and atherosclerosis due to endothelial dysfunction, dyslipidaemia and platelet activation.15,22,23 In this editorial I will briefly discuss the VESUVIUS study, which sought to add new data to the limited body of evidence pertaining to the vascular effects of e-cigarettes.9
Table 2. Potential cardiovascular effects of e-cigarettes components 12-19 | |
Effect | Component potentially involved |
Increased blood pressure | Acrolein, PMs, nicotine |
Myocardial dysfunction and cardiomyopathy | Acrolein |
Reduced cardiac contractility | Acrolein, PMs |
Increased risk of ventricular arrhythmias | Acrolein, PMs |
Impaired vascular repair and vascular injury | Acrolein |
Increased risk of thrombosis | Acrolein, PMs |
Coronary artery disease and myocardial infarction | PMs, nicotine |
PM ultrafine particles. | |

Figure 2. Diagram summarising the potential cardiovascular effects of e-cigarettes12-23
The VESUVIUS study
In this 2019 Journal of American College of Cardiology paper by George and colleagues9 the authors sought to examine the early vascular effects of switching from tobacco cigarettes to e-cigarettes. The VESUVIUS (vascular effects of regular cigarette versus electronic cigarette use) study was a British Heart Foundation-commissioned prospective randomised controlled study conducted between August 2016 and July 2018 in a single tertiary centre.
Design and methods
The study included a parallel, non-randomised preference cohort and blinded endpoint of smokers aged 18 years or above who had smoked at least 15 cigarettes per day for at least 2 years. Inclusion and exclusion criteria are in Table 3. The study was undertaken from August 2016 to July 2018 in a single tertiary research centre. Participants were recruited from local advertisements, smoking cessation databases, and visits to local businesses, as well as via the Scottish Primary Care Research Network.
Table 3. Inclusion and exclusion criteria for the VESUVIUS study9 | |
|---|---|
Inclusion
| Exclusion |
Patients aged 18 years or more
| Patients aged <18 years |
Minimum smoking history of 15 or more cigarettes a day for a minimum of 2 years
| Established cardiovascular disease Diabetes Chronic kidney disease On treatment for the above conditions |
Consented candidates in agreement to stop smoking were randomised to one of the e-cigarettes (EC) arms in a 1:1 ratio using a web-based randomization system to either: a) e-cigarette containing 16 mg nicotine (EC-nicotine) or b) nicotine-free e-cigarette plus nicotine flavouring (EC-nicotine-free). Participants unwilling to stop smoking continued in the parallel preference tobacco cigarette (TC) cohort. A propensity score analysis was carried out to adjust for differences between the randomised and preference arms. Vascular function was obtained from flow mediated dilation (FMD) and pulse wave velocity. These vascular outcomes have been shown to be predictors of cardiovascular risk.10,24
Endothelial function was assessed by measuring FMD of the brachial artery using ultrasound at baseline and at one month. Pulse waved velocity (PWV) was measured at baseline and 1 month by a single operator blinded to study assignment. The operator measuring FMD and PWV was the same. It was not specified from the paper whether PWV was measured using the radial or brachial artery, although the mention of the SphygmoCor used would suggest that the measurements were brachial. In addition, biomarkers such as low-density lipoprotein and high sensitivity C-reactive protein were measured. Compliance with EC was measured by carbon monoxide levels.
The authors hypothesised that nicotine may accelerate the atherogenic process by binding to high-affinity nicotinic acetylcholine receptor cell surface receptors.9,20 However, longer-term nicotine use appears not to accelerate atherogenesis but may contribute to acute cardiovascular events in the presence of cardiovascular disease.9,21 In addition, the authors’ assumption was that improvement in FMD could lead to reduced cardiovascular events.10
Endpoints
The primary endpoint was change in FMD among the TC group and the EC-nicotine and EC-nicotine-free groups. The primary outcome was described as the change in FMD among the TC group and the EC-nicotine and EC-nicotine free arms. Secondary outcomes included change in FMD, PWV, augmentation index at 75 beats/min, heart rate, blood pressure, and biomarkers for the TC, EC-nicotine, and EC-nicotine-free arms.
Results
A total of 145 participants were recruited into the study. Of these, 114 patients (40 TC, 37 EC-nicotine, 37 EC-nicotine-free) completed both visits. Baseline demographics appeared well-matched amongst the various groups (see Table 4).
Table 4. Baseline demographics of the VESUVIUS study9 | |||
|---|---|---|---|
TC | EC-Nicotine | EC-Nicotine-Free | |
Participants, n (%) | 40 (35) | 37 (32.5) | 37 (32.5) |
Male | 13(32.5) | 14 (37.8) | 12 (33.4) |
Age, years | 44.2 (0.4-47.9) | 48.0 (44.7-51.3) | 48.4 (43.5–53.3) |
Body mass index | 26.7 (25.0–28.5) | 28.1 (25.8–30.4) | 27.1 (25.4–28.8) |
Cigarettes/ day | 20 [15-20] | 18 [15-20] | 18 [15-20] |
Years smoked | 29.0 [19.5–36.5] | 36.0 [25.0–41.0] | 32.0 [22.0–40.0] |
Pack year history | 25.4 [15.5–36.5] | 33.3 [21.8–44.0] | 27 [19.9–36.8] |
Values expressed as n (%), mean (95% confidence interval), or median [interquartile range]. EC electronic cigarette, TC tobacco cigarette. | |||
Primary outcomes
FMD of the brachial artery showed a significant trend in the difference among arms from TC to EC-nicotine to EC-nicotine-free (linear trend b for TC, EC-nicotine, EC-nicotine-free 0.73%, 95% CI 0.41-1.05; p<0.0001). Within 1 month of switching from TC to EC, FMD significantly improved among TC and combined EC arms (1.49%, 95% CI 0.93-2.04; p<0.0001), as well as separately between TC and EC-nicotine and between TC and EC-nicotine-free. There was no statistically significant difference in FMD change between the EC-nicotine and EC-nicotine-free arms. In subgroup analyses, the improvement in vascular function was significantly greater in women who switched from TC to EC.
Secondary outcomes
There was no significant trend in difference among the 3 arms for other secondary outcomes including PWV, heart rate, and biomarkers of inflammation and platelet reactivity.9 However, as there were significant interaction terms for PWV between the treatment and smoking pack years, a subgroup analysis was carried out. This revealed that smokers that had 20 or less pack-years demonstrated an improvement from TC to EC with an improvement in PWV (-0.471m/s, 95% CI -0.834 to -0.107; p=0.014), whereas those who smoked >20 pack years showed no change within this time-frame.
Author’s conclusions
The authors concluded that switching from TC to EC may be considered a harms reduction measure, with females showing a larger improvement in vascular health within one month of changing from TC to EC. However, they also state that despite their findings, e-cigarettes should not be viewed as harmless as there was no evidence demonstrating that EC are safe per se.
Discussion
The VESUVIUS study showed a clear benefit in vascular function when switching from tobacco smoking to e-cigarettes, These findings arein keeping with Ikonomidis et al who demonstrated a worsening in oxidative stress and arterial elasticity among tobacco smokers compared to e-cigarette users.11 The VESUVIUS study was well executed and designed; it was a longitudinal study design with discontinuation of tobacco in two study groups. In particular the authors measured compliance, which is a key cofounding factor in lifestyle modification studies. The fact that FMD and PWV were both measured by the same operator also reduced the risk of operator variability, and the propensity score as adjustment covariate allowed control of potential biases.
There were, however a number of limitations. FMD and PWV remain soft endpoints, although often utilized because there is evidence showing that they are associated with cardiovascular risk.10,24 The assumption is that in time these improvements will translate into a clinical benefit and reduction in cardiovascular events. However, one important limitation of this study is that there was no non-smoking control group. Moving forward with research it would be interesting to compare non-smokers with those who switched from tobacco to e-cigarettes, as this study does not answer whether e-cigarette consumption is associated with endothelial dysfunction. As the actual FMD values were not published in this study we were unable to compare with values from previous published studies of healthy/elderly individuals.
It is also interesting to note that there were no statistical differences between EC-nicotine and EC-nicotine free, which would suggest that nicotine is not responsible for endothelial dysfunction. This is contrary to previous a previous study by Neunteufl, 25 which demonstrated that nicotine alone causes acute endothelial dysfunction although to a lesser extent than smoking a cigarette of the same nicotine yield.
Other limitations include the small sample size and short follow-up time period. For instance, it would be important to evaluate whether the difference in endothelial function seen is sustained over a longer timeframe; and also whether males do benefit to the same extent as female in the long term.
Conclusion
The VESUVIUS study was a well conducted study demonstrating clear benefit in endothelial function in tobacco smokers just one month after switching to e-cigarette use. However larger studies with longer follow up are required to evaluate whether these benefits translate into better cardiovascular outcomes. The improvements in endothelial function in this study are likely relate to tobacco discontinuation rather than a beneficial effect of e-cigarettes. While e-cigarettes may be a useful adjunct for smoking cessation in tobacco smokers, their use should not be encouraged in other settings (e.g. new smokers) until more safety data is available.
Disclosures
None.
- World Health Organization. WHO Report on the Global Tobacco Epidemic, 2008: The MPOWER Package. Geneva, Switzerland: World Health Organization: 2008. Available at: http://www.who.int/tobacco/mpower/mpower_report_full_2008.pdf. Accessed 23rd Dec 2019.
- American Cancer Society. Harmful chemicals in tobacco products. Available at: https://www.cancer.org/cancer/cancer-causes/tobacco-and-cancer/carcinogens-found-in-tobacco-products.html. Accessed 17th Dec 2019.
- Qasim H, Karim Z, Rivera J et al. Impact of Electronic Cigarettes on the Cardiovascular System. Journal of the American Heart Association. 2017 Aug; 6(9):1-14
- Orellana-Barrios M; Payne D; Mulkey Z, et al. (2015). Electronic cigarettes-a narrative review for clinicians. The American Journal of Medicine. 2015; 128 (7): 674–81.
- E-cigarettes/vapes. Smokefree NHS website. Available at: https://www.nhs.uk/smokefree/help-and-advice/e-cigarettes.
- ASH (Action on smoking and Health). Use of e-cigarettes (vaporisers) among adults in Great Britain. Available at: https://ash.org.uk/wp-content/uploads/2019/09/Use-of-e-cigarettes-among-adults-2019.pdf. Accessed 22nd December 2019.
- Alzahrani T, Pena I, Temesgen N, Glantz SA. Association Between Electronic Cigarette Use and Myocardial Infarction [published correction appears in Am J Prev Med. 2019 Oct;57(4):579-584]. Am J Prev Med. 2018;55(4):455–461. doi:10.1016/j.amepre.2018.05.004
- Christiani D. Vaping-induced acute lung injury. N Engl J Med. 2020 Mar; 382:960-962
- George J, Hussain M, Vadiveloo T, et al. Cardiovascular effects of switching from tobacco cigarettes to electronic cigarettes. J AM Coll Cardiol. 2019; 74(25): 3112-3120.
- Inaba Y., Chen J.A., Bergmann S.R. Prediction of future cardiovascular outcomes by flow-mediated vasodilatation of brachial artery: a meta-analysis. Int J Cardiovasc Imaging. 2010; 26: 631–640.
- Ikonomidis I, Vlastos D, Kourea K et al. Electronic Cigarette Smoking Increases Arterial Stiffness and Oxidative Stress to a Lesser Extent Than a Single Conventional Cigarette: An Acute and Chronic Study. Circulation. 2018;137(3): 303-306
- Qasim H, Karim ZA, Rivera JO, Khasawneh FT, Alshbool FZ. Impact of Electronic Cigarettes on the Cardiovascular System. J Am Heart Assoc. 2017 Aug 30;6(9):e006353. doi: 10.1161/JAHA.117.006353. PMID: 28855171; PMCID: PMC5634286.
- Perez CM, Hazari MS, Ledbetter AD, Haykal‐Coates N, Carll AP, Cascio WE, Winsett DW, Costa DL, Farraj AK. Acrolein inhalation alters arterial blood gases and triggers carotid body‐mediated cardiovascular responses in hypertensive rats. Inhalation Toxicol. 2015;27:54–63.
- Hazari MS, Haykal‐Coates N, Winsett DW, Krantz QT, King C, Costa DL, Farraj AK. TRPA1 and sympathetic activation contribute to increased risk of triggered cardiac arrhythmias in hypertensive rats exposed to diesel exhaust. Environ Health Perspect. 2011;119:951–957.
- Wheat LA, Haberzettl P, Hellmann J, Baba SP, Bertke M, Lee J, McCracken J, O'Toole TE, Bhatnagar A, Conklin DJ. Acrolein inhalation prevents vascular endothelial growth factor‐induced mobilization of Flk‐1+/Sca‐1+ cells in mice. Arterioscler Thromb Vasc Biol. 2011;31:1598–1606
- Sithu SD, Srivastava S, Siddiqui MA, Vladykovskaya E, Riggs DW, Conklin DJ, Haberzettl P, O'Toole TE, Bhatnagar A, D'Souza SE. Exposure to acrolein by inhalation causes platelet activation. Toxicol Appl Pharmacol. 2010;248:100–110
- Peters A, Dockery DW, Muller JE, Mittleman MA. Increased particulate air pollution and the triggering of myocardial infarction. Circulation. 2001;103:2810–2815.
- Sullivan J, Sheppard L, Schreuder A, Ishikawa N, Siscovick D, Kaufman J. Relation between short‐term fine‐particulate matter exposure and onset of myocardial infarction. Epidemiology. 2005;16:41–48.
- Wang T, Lang GD, Moreno‐Vinasco L, Huang Y, Goonewardena SN, Peng YJ, Svensson EC, Natarajan V, Lang RM, Linares JD, Breysse PN, Geyh AS, Samet JM, Lussier YA, Dudley S, Prabhakar NR, Garcia JG. Particulate matter induces cardiac arrhythmias via dysregulation of carotid body sensitivity and cardiac sodium channels. Am J Respir Cell Mol Biol. 2012;46:524–531.
- Heeschen C., Jang J.J., Weis M., et al. (2001) Nicotine stimulates angiogenesis and promotes tumor growth and atherosclerosis. Nat Med 7:833–839.
- Benowitz N.L., Burbank A.D. (2016) Cardiovascular toxicity of nicotine: implications for electronic cigarette use. Trends Cardiovasc Med 26:515–523
- Srivastava S, Sithu SD, Vladykovskaya E, Haberzettl P, Hoetker DJ, Siddiqui MA, Conklin DJ, D'Souza SE, Bhatnagar A. Oral exposure to acrolein exacerbates atherosclerosis in apoE‐null mice. Atherosclerosis. 2011;215:301–308.
- Conklin DJ, Barski OA, Lesgards JF, Juvan P, Rezen T, Rozman D, Prough RA, Vladykovskaya E, Liu S, Srivastava S, Bhatnagar A. Acrolein consumption induces systemic dyslipidemia and lipoprotein modification. Toxicol Appl Pharmacol. 2010;243:1–12.
- Maruhashi T, Soga J, Fujimura N, et al. Endothelial Dysfunction, Increased Arterial Stiffness, and Cardiovascular Risk Prediction in Patients with Coronary Artery Disease: FMD-J (Flow-Mediated Dilation Japan) Study A. J Am Heart Assoc. 2018;7(14):e008588. doi:10.1161/JAHA.118.008588
- Neunteufl T, Heher S, Kostner K, et al. Contribution of nicotine to acute endothelial dysfunction in long-term smokers. J Am Coll Cardiol. 2002;39(2):251–256. doi:10.1016/s0735-1097(01)01732
Community Events Calendar


 a
a b
b c
c d
d