Magnetic Resonance Imaging in Patients with Cardiac Implantable Electronic Devices: Current Recommendations
| Take Home Messages |
|---|
|
Cardiac Implantable Electronic Devices (CIED) comprise loop recorders (ILR), pacemakers (PPM), defibrillators (ICD) and cardiac re-synchronisation therapy (CRT). It is estimated there are >1,000,000 people with a cardiac device worldwide.(1) Within the last two decades, CIED were often a contraindication to Magnetic Resonance Imaging (MRI).(2) Some doctors may be unaware that with technological developments and sufficient preparation, MRI is now commonly very safe for this patient group. One concern is that patients with CIED may now be receiving suboptimal care due to the historical restriction surrounding MRI. In 2017 the Heart Rhythm Society (HRS) released a comprehensive statement on the safe use of MRI in patients with CIED.(3) This editorial summarises the statement in the hopes of increasing the acceptability and use of MRI for those with a CIED.
Basic Concepts
MRI is based on the properties of hydrogen nuclei absorbing and emitting radiofrequency energy when exposed to a magnetic field.⁴ The scanner can create static and pulsed magnetic fields, and field gradients to generate images.
An MRI scanner commonly operates between 1.5-3 Tesla (T=magnetic density). The force generated is >60,000x the strength of the Earth’s magnetic field and can create serious interference with metallic objects. The magnet is always ‘on’ but the further away from the scanner the less its magnetic effect. The MRI environment is divided into four zones of safety. Zone 4 comprises the MRI scan room; where the magnetic effects are greatest and no metal (including resuscitative equipment) can enter this zone. Anyone with any metallic implants or equipment should consult a member of MRI staff before entering zone 2 and above due to the risk of magnetic interference. Figure 1 demonstrates the typical MRI environment as outlined by the American College of Radiology.
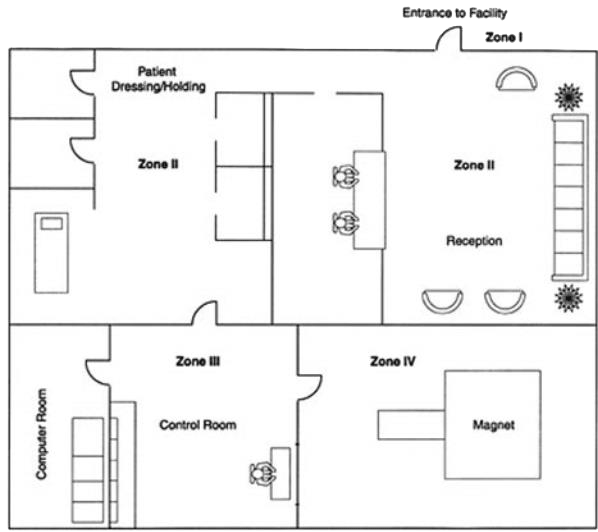
Figure 1: Typical safety zones within the MRI environment where zone 4 holds the greatest risk of magnetic interference⁵
Conditional and Non-Conditional Devices
No CIED can be termed “MRI Safe”. The term “MRI conditional” implies that MR scanning can take place without hazard if certain “conditions” are fulfilled. These conditions include:
- Using makes and models of generator and leads (or a combination thereof) that have been designed, tested and confirmed to function safely in MRI
- No fractured, redundant or epicardial leads are present
- Appropriate checks and programming of device pre and post scan
- Safety protocol followed which comprises conduct of the scanner, appropriately trained personnel and patient monitoring
Device manufacturers hold records of which devices and leads (combinations thereof) have been given conditional status. They may also update information on historic leads so it is worth checking with the manufacturer if you require further information about a patient’s system.
The label ‘MRI non-conditional’ will apply to any system/combination that does not meet the criteria above. It should be noted it is usually safe to scan someone with an MR nonconditional device; several studies have been performed without significant adverse events in non-conditional devices.(3,6,7) HRS therefore recommends that MRI in non-conditional devices can take place following appropriate preparation (as long as no abandoned leads are present).
Potential Effects of MRI on CIED
Below are the potential hazards of MRI with CIED. Although they would be uncommon in adequately programmed contemporary devices, some effects may be more likely to occur in non-conditional systems or the inadequately programmed device.
Field-Induced Force & Torque
CIED leads do not contain sufficient ferrous material to be moved by MRI scanning. The device generators (including ILR) are confined by subcutaneous tissue and are unlikely to move. They will create some surrounding image artefact which may interfere with overall quality.
Field-Induced Electrical Current
The MRI can create currents within the leads which can cause myocardial capture and atrial or ventricular arrhythmia. Electromagnetic pulses may cause over-sensing and artefact which can impact demand pacing or inhibit pacing. It may also trigger tachycardia therapies.
Heating & Tissue Damage
MRI can create thermal damage at the lead tip causing a type of functional ablation. This may alter sensing and capture thresholds which may not be immediately detectable. Devices should be checked within six months post scan to check no significant change in threshold has occurred. Non-conditional systems may experience a higher degree of heating and a greater rise in threshold. It is therefore recommended to check the threshold at an earlier interval in these patients.
Reed Switch
The reed switch is a component designed to enable interrogation of the device when the magnetic programming header is in contact.(8) The reed switch will trigger a certain response (depending on manufacturer specifications) when exposed to a magnetic field. For example, when exposed to a magnet, ICDs will inhibit tachycardia therapy. Pacemakers will have different responses, but may deliver asynchronous pacing at high outputs.
Electrical Reset
A rare but serious effect is if a power-on electrical reset is triggered. Pacing may be inhibited and tachycardia therapy activated. Pacing may be delivered below the required pacing threshold, battery function may be affected and programming the device may be limited for a time. The device may not respond to external magnet application in the usual fashion. Different manufacturers have details of how the device will default if power-on reset is triggered by MRI environment and a summary is available in the HRS statement.(3) In these instances, external treatment of any arrhythmia may be required until reset mode terminates.
Battery
Magnetic interference or prior programming may result in high output asynchronous pacing throughout the scan. Current drain during MRI can be significant and devices that were nearing elective replacement indicator (ERI) may drain sufficiently to enter this battery protection mode during the scan.(8) In ERI mode there may be limits on how the device can be subsequently interrogated and programmed. It is therefore advised that patients nearing ERI have their generator changed prior to having an MRI performed.
The HRS have devised a checklist to ensure safe conduct of MRI in those with CIED.(3) This is summarised as:
- Responsible physician present (staff able to programme CIED required in pacing dependent and non-conditional patients)
- Monitor ECG and O2 saturations
- Defibrillator with external pacing function available
- ALS trained staff available
- CIED interrogation before and after MRI
- ICD - tachycardia detection and treatment algorithms deactivated
- Follow up takes place within 3-6 months post scan
Loop Recorders
MRI up to 3 Tesla has been described as safe in patients with ILR, however it is recommended to interrogate the device before the scan to save any relevant data. The MRI field is likely to generate artefact which will be stored on the device. Therefore interrogating the ILR and clearing its data post-scan is also recommended.
Redundant, Fractured or Epicardial Leads
Although trials have shown a negligible heating effect in abandoned leads,(9) which doesn’t appear to cause any clinical sequelae.(10) The HRS currently advise that MRI is avoided in patients with abandoned, epicardial or damaged leads due to paucity of data and uncertain risk to patients.
Table 1 demonstrates the key aspects of the safety protocol to follow for any patient with a CIED undergoing MRI scanning.
| At time of MRI Request | Pre-Scan | Post-Scan |
|---|---|---|
| Rule out any fractured or abandoned leads | Note current device settings and programme for safe MR behaviour | Check and restore usual settings |
| Gather device and lead information to establish if MR conditional system | Store any relevant arrhythmia logs | Clear any artifact data stored |
| Establish if pacing dependent | Ensure appropriate equipment and personnel available | Arrange device follow up within 3-6 months (within 1 week for non-conditional systems) |
| Check battery, arrhythmia logs, lead parameters |
Table 1: Summary of safety steps required before and after scanning
Table 2 indicates how best to prepare patients for the scan depending on whether an intrinsic rhythm is present or not.
| Pacing Dependent | Non-Dependent |
|---|---|
| Physician able to temporarily pace available | Programme to non-pacing mode (0V0, 0D0), or |
| Personnel able to programme CIED in attendance | Programme to inhibited mode (VVI/DDI) with deactivation of adaptive features |
| Programme asynchronous pacing mode | If CRT programme to asynchronous pacing with rate set to avoid competitive pacing |
| Deactivate rate adaptive features and set rate to avoid competitive pacing |
Table 2: Safely preparing for MRI in pacing dependent and non-dependent patients
In the event that either a patient enters the scanner without appropriate CIED preparation, or MRI-CIED interference occurs; below are the emergency measures that should be undertaken if arrhythmia develops during the scan.
Emergency Measures: Bradycardia during MRI
- Abort scan if even transient bradycardia or inhibition of pacing is noted
- Remove patient from zone 4 (scan room)
- If pacing has not resumed
- Apply magnet to device
- Programme the device to asynchronous pacing
- If unable to programme/no magnet response; commence external/temporary pacing
Emergency Measures: Tachycardia during MRI
- Abort scan if sustained ventricular arrhythmia occurs
- Remove patient from zone 4 (scan room)
- Re-programme tachycardia therapies 'ON'
- Commence adult life support and external cardioversion as required
Streamlining the Process
Many sites may still encounter difficulties when attempting to arrange an MRI for a patient with a CIED. One centre, having developed a streamlined protocol, saw a twenty-fold increase in numbers of patients with CIED receiving MRI as a result.¹¹ To achieve this they used pre-allocated sessions for CIED scanning, thus ensuring the necessary staff were available. They delivered training to this team which included administrators, physicians, radiographers, and physiologists to improve cross-team working, minimize unnecessary scan refusals and increase efficiency and confidence. Scanning is performed using lower specific absorption rate power depending on manufacturer guidelines and devices are programmed as outlined above. By creating a standardised proforma, device details could be checked at time of booking and patients then allocated to the dedicated CIED session. Standard operating procedures, further information and guidelines can be found on www.mrimypacemaker.com.
Conclusion
The HRS has released a consensus statement offering a recommended checklist and protocol that can be followed for patients with CIED requiring MRI.(3) The majority of modern CIED will be MRI conditional systems and can undergo MRI if safety conditions are met. Conditions include certain model/s and combination of generator and leads used, adequate device checks and programming pre and post scan, appropriately trained personnel being available and no redundant or epicardial leads being present. By pre-allocating patients with CIED to an MRI session where the necessary staff and equipment are available, a more efficient process can be developed and uptake of MRI could be increased.
Non-conditional devices can also undergo scanning but additional staff and checks are required and the scan will be conducted with an acknowledgement that potential, unpredictable risk may exist for the patient. At present MRI is not recommended in those with abandoned, damaged or epicardial leads.
It is hoped the general cardiologist will feel sufficiently informed of the safety of MRI in those with a CIED and ultimately increase patient access to MRI as clinically required. You may wish to update other colleagues in your region that MRI should be offered to the majority of patients with CIED where clinically indicated.
References
- Wood MA, Ellenbogen K. Cardiac pacemakers from the patient's perspective. Circulation. 2002;105(18):2136-8.
- Mitka M. First MRI-Safe Pacemaker Receives Conditional Approval From FDA. JAMA. 2011;305(10):985-6.
- Julia H. Indik, Rod Gimbel, Haruhiko Abe, et al. 2017 HRS expert consensus statement on magnetic resonance imaging and radiation exposure in patients with cardiovascular implantable electronic devices. Heart Rhythm Society; 2017.
- Berger A. Magnetic resonance imaging. BMJ. 2002;324(7328):35.
- Kanal E, Barkovich AJ, Bell C, et al. ACR guidance document on MR safe practices: 2013. Journal of Magnetic Resonance Imaging. 2013;37(3):501-30.
- Eyvazian V, Do DH, Bayoneta AJ, et al. Safety of Cardiac Magnetic Resonance Imaging with Non-Conditional Cardiac Implantable Electronic Devices Journal of the American College of Cardiology. 2017;69(11):429-.
- Sommer T, Naehle CP, Yang A, et al. Strategy for safe performance of extrathoracic magnetic resonance imaging at 1.5 tesla in the presence of cardiac pacemakers in non-pacemaker-dependent patients: a prospective study with 115 examinations. Circulation. 2006;114(12):1285.
- Irnich W, Irnich B, Bartsch C, et al. Do we need pacemakers resistant to magnetic resonance imaging? Europace : European pacing, arrhythmias, and cardiac electrophysiology : journal of the working groups on cardiac pacing, arrhythmias, and cardiac cellular electrophysiology of the European Society of Cardiology. 2005;7(4):353-65.
- Langman DA, Goldberg IB, Finn JP, et al. Pacemaker lead tip heating in abandoned and pacemaker-attached leads at 1.5 Tesla MRI. Journal of magnetic resonance imaging : JMRI. 2011;33(2):426-31.
- Higgins J, Gard J, Espinosa R, et al. Safety and Outcomes of Magnetic Resonance Imaging in Patients With Abandoned Pacemaker and Defibrillator Leads. Circulation. 2013;128(22).
- Bhuva AN, Feuchter P, Hawkins A, et al. MRI for patients with cardiac implantable electronic devices: simplifying complexity with a ‘one-stop’ service model. BMJ Quality & Safety. 2019.
Community Events Calendar


