Atrial fibrillation - a ‘large’ problem?
| Take Home Messages |
|---|
|
Introduction
Atrial fibrillation (AF) is the most common sustained arrhythmia and is associated with an increased risk of stroke, heart failure and death.(1,2) AF is a significant public health issue with an estimated global incidence of 5 million new cases per year(3) and this number is expected to rise in the future.(4) By 2060, it is predicted that a total of 17.9 million people in Europe will be affected by the condition.(5) In addition to an ageing population, trends of rising body-mass index (BMI) are thought to contribute to the growing AF epidemic. A global population-based study found an increase in mean BMI over the past four decades from 21.7 kg/m2 to 24.2 kg/m2 in men and from 22.1 kg/m2 to 24.4 kg/m2 in women.(6) Over the same time period, the age-standardised prevalence of obesity increased from 3.2% to 10.8% in men and 6.4% to 14.9% in women. Therefore, we cannot afford to ignore this ‘large’ problem in the management of patients with AF. In this editorial, I will briefly outline the impact of obesity on the pathophysiology, outcomes and management of AF. Further, I will describe how a structured weight loss and risk factor modification approach can help improve outcomes in obese patients with AF.
Pathophysiology of obesity in AF
Obesity has consistently been shown to increase the risk of incident AF.(7–9) In fact, a meta-analysis of ten studies by Jones et al. (2019) demonstrated that the incidence of AF increased by 13% for every 5% gain in weight.(9) The underlying pathophysiological mechanisms linking excess weight and AF are poorly understood but may be related to left ventricular hypertrophy, diastolic dysfunction, left atrial dilatation, increased atrial fibrosis and epicardial adipose tissue.(10) In addition, obesity is an independent risk factor for other cardiovascular diseases (e.g. hypertension, coronary artery disease, metabolic syndrome) that in turn increase the risk of AF. Another important condition that correlates excess weight with AF is obstructive sleep apnoea which is more common in obesity and increases the risk of AF by 4-fold.(11)
Atrial fibrillation causes electrophysiological, mechanical and structural re-modelling that creates a substrate within the atria for further arrhythmia.(12) As such, the term ‘AF begets AF’ is often used. The natural progression of the disease is such that many patients who initially suffer from paroxysmal AF will eventually progress to persistent AF. Progression of the disease appears to be accelerated in patients with a raised BMI.(13) It was also observed that there was a graded relationship between BMI and progression of the disease, with those at the highest BMI being most at risk.
Many patients with AF are asymptomatic or have very minor symptoms. As a result, it can remain undiagnosed for a long period of time. However, in those with the condition, increasing BMI was associated with worsening AF symptom severity, burden and quality of life(14) (Figure 1). The study by Chalazan et al. (2018) used the Toronto Atrial Fibrillation Severity Scale (AFSS) to assess the severity of AF.
The AFSS score is comprised of three categories:
- AF symptoms (palpitations, chest pain, dyspnoea, exercise intolerance, fatigue, dizziness);
- AF burden (frequency, duration, first episode severity, last episode severity); and
- Quality of life (life assessment, cardioversion, emergency visits, admissions, specialist visits).
Each parameter is rated by the patient (from 0 to 5) and the sum of ratings used to calculate the total AFSS score (max 35 points) with higher scores indicating greater AF severity. The authors demons- -trated that patients with a raised BMI were more symptomatic of AF, regardless of whether they were treated with a rate or rhythm control strategy (including catheter ablation).
Impact of obesity on AF management
The management of AF involves initiation of anticoagulation therapy in those at high risk of stroke, rate and/or rhythm control and management of risk factors and concomitant conditions.(15) There has been no definitive evidence to suggest a difference in the effectiveness of a rate control strategy among those with a raised BMI. However, in contrast, rhythm control appears to be less efficacious in obese patients with AF and associated with higher risk of procedural harm.(16) In a study of 752 AF patients treated with anti-arrhythmic medications, Vinolas et al. (2013) reported that obesity was independently associated with a lower probability of pharmacological reversion to sinus rhythm.(17) Furthermore, raised BMI was also linked to a higher rate of AF recurrence at 1-year following catheter ablation(18) and higher risk of complications following catheter ablation.(16)
In another study of 125 AF patients undergoing direct current cardioversion (DCCV), the routine use of 200 J was found to be inadequate in obese patients, with higher success observed using paddles and escalation to 360 J.(19)
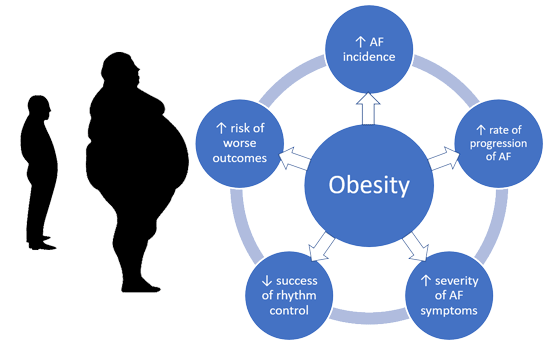
Figure 1: The effects of obesity in patients with atrial fibrillation
Whilst shock vector (anterior-posterior compared to antero-apical) had no impact on DCCV success, using the manual pressure augmentation (MPA) technique improved chances of success. The MPA technique involves the application of manual pressure during the expiratory phase of respiration, by either one or two operators wearing latex gloves using either one or two hands, while another clinician charged and delivered the shock through the defibrillator. The MPA technique was successful in 80% of patients who failed both patches and paddles.
Anticoagulation in obese AF patients
Anticoagulation using either a vitamin K antagonist (e.g. warfarin) or a non-vitamin K antagonist oral anticoagulant (NOAC) is recommended in AF patients at high risk of stroke.(15) There were initial concerns about possible reduced efficacy with the use of fixed-dose NOACs in patients with raised BMI. However, data from large randomised controlled and registry-based studies to investigate this have been reassuring. In fact, two systematic reviews have surprisingly shown better outcomes in overweight and obese patients treated with a NOAC compared to those with normal or low body weight leading to the term ‘BMI paradox’.(20,21) Although the reasons for this observation remain unclear, the benefits of higher body weight seem to be driven by improved efficacy as no significant differences in bleeding risk were observed. Additionally, greater benefits were seen with factor Xa inhibitors (Rivaroxaban, Apixaban and Edoxaban) compared to the direct thrombin inhibitor (Dabigatran). Analysis of real world registry data suggested that obese patients receiving factor Xa inhibitors had lower rates of stroke whilst the direct thrombin inhibitor was associated with a higher rate of stroke/venous thromboembolism in patients with a body weight >100 kg.(21)
Structured weight loss in obese AF patients
Despite the negative effects of obesity in AF (Figure 1), there is perhaps still hope for patients with both these conditions. The LEGACY trial by Pathak et al. (2015) recruited 1,415 consecutive patients with AF and were able to demonstrate that long-term sustained weight loss was associated with a significant reduction in AF burden and an increased chance of maintenance of sinus rhythm.(22) Patients with at least 10% weight loss had a 6- fold greater probability of arrhythmia-free survival compared to those who were unable to achieve this target. In the subsequent REVERSE-AF trial, the authors demonstrated a dynamic relationship between weight and AF using strict weight-loss and risk factor management (Table 1).(23) Patients in the study were categorised into three groups according to the amount of weight loss: Group 1 (<3%), Group 2 (3-9%) and Group 3 (≥10%). During the follow-up period of four years, there were a total of 41%, 26% and 3% of patients from Group 1, Group 2 and Group 3, respectively, who progressed from paroxysmal to persistent AF and a total of 26%, 49% and 88% of patients from Group 1, Group 2 and Group 3, respectively, who reversed from persistent to paroxysmal or no AF.
Table 1: Example of a weight-loss and risk factor management programme adapted from the REVERSE-AF trial(24)
| Weight-loss management | |
|---|---|
| Intervention | Description |
| Clinic review | Dedicated physician-led clinic focused on weight and risk factor management. |
| Individualised programme | Goal-directed, motivational, structured programme with one-to-one counselling. |
| Journal keeping | Each patient maintained a personal lifestyle journal. |
| Meal plan | Each patient was provided with a meal plan targeting an initial weight loss>10 %. |
| Meal replacement | Patients who lost <3 % weight at 3 months were prescribed a meal replacement; or those who achieved the initial weight loss of >10 % were prescribed high protein, low glycaemic index, calorie-controlled food to achieve a target body mass index of ≤25 Kg/m2. |
| Exercise | Advised to undertake 30 mins of exercise, 3-4 times per week with an aim to increase to a total of 200 mins per week. |
| Risk factor management | |
| Risk factor | Screening/intervention |
| Hypertension | Salt restriction Pharmacotherapy (as required) Home blood pressure monitoring 2-3 times per day, with a target for 80 % of recordings to be <130/80 mmHg Office blood pressure monitoring Exercise stress testing for screening of exercised-induced hypertension 24-hour ambulatory blood pressure monitoring Echocardiography to evaluate left ventricular hypertrophy |
| Dyslipidaemia | Lifestyle measures Pharmacotherapy (as required) |
| Glucose intolerance | Lifestyle measures Pharmacotherapy (as required) |
| Obstructive sleep apnoea | Sleep study Continuous positive airway pressure if apnoea-hypopnoea index ≥30/hour |
| Smoking | Advised on smoking cessation |
| Excess alcohol | Advised to reduce alcohol to ≤30 g/week |
Conclusions
AF and obesity are commonly encountered in the clinical setting. Patients with excess weight are at increased risk of incident AF, accelerated progression of the disease, more severe symptoms, reduced likelihood of effective treatment and higher rates of complications. Accumulating evidence demonstrates that risk factor management including a structured weight-loss programme in obese AF patients can improve outcomes.
References
- Benjamin EJ, Wolf PA, D’Agostino RB, et al. Impact of atrial fibrillation on the risk of death: the Framingham Heart Study. Circulation 1998;98:946–52.
- Stewart S, Hart CL, Hole DJ, et al. A population-based study of the long-term risks associated with atrial fibrillation: 20-year follow-up of the Renfrew/Paisley study. Am J Med 2002;113:359–64.
- Chugh SS, Havmoeller R, Narayanan K, et al. Worldwide epidemiology of atrial fibrillation: a Global Burden of Disease 2010 Study. Circulation 2014;129:837-47.
- Morillo CA, Banerjee A, Perel P, et al. Atrial fibrilation: the current epidemic. J Geriatr Cardiol 2017;14: 195–203.
- Krijthe BP, Kunst A, Benjamin EJ, et al. Projections on the number of individuals with atrial fibrillation in the European Union, from 2000 to 2060. Eur Heart J 2013; 34:2746–51.
- Trends in adult body-mass index in 200 countries from 1975 to 2014: a pooled analysis of 1698 populationbased measurement studies with 19.2 million participants. Lancet (London, England) 2016;387:1377–96
- Asad Z, Abbas M, Javed I, et al. Obesity is associated with incident atrial fibrillation independent of gender: A meta-analysis. J Cardiovasc Electrophysiol 2018;29:725-32.
- Berkovitch A, Kivity S, Klempfner R, et al. Body mass index and the risk of new-onset atrial fibrillation in middle-aged adults. Am Heart J 2016;173:41–8.
- Jones NR, Taylor KS, Taylor CJ, et al. Weight change and the risk of incident atrial fibrillation: a systematic review and meta-analysis. Heart Published Online First: June 2019.
- Goudis CA, Korantzopoulos P, Ntalas I V, et al. Obesity and atrial fibrillation: A comprehensive review of the pathophysiological mechanisms and links. J Cardiol 2015;66:361–9.
- Mehra R, Benjamin EJ, Shahar E, et al. Association of nocturnal arrhythmias with sleep-disordered breathing: The Sleep Heart Health Study. Am J Respir Crit Care Med 2006;173:910–6.
- Wijesurendra RS, Casadei B. Mechanisms of atrial fibrillation. Heart Published Online First: August 2019.
- Tsang TSM, Barnes ME, Miyasaka Y, et al. Obesity as a risk factor for the progression of paroxysmal to permanent atrial fibrillation: a longitudinal cohort study of 21 years. Eur Heart J 2008;29:2227–33.
- Chalazan B, Dickerman D, Sridhar A, et al. Relation of Body Mass Index to Symptom Burden in Patients with Atrial Fibrillation. Am J Cardiol 2018;122:235–41.
- Kirchhof P, Benussi S, Kotecha D, et al. 2016 ESC Guidelines for the management of atrial fibrillation developed in collaboration with EACTS. Eur Heart J 2016;37:2893–962.
- Shoemaker MB, Muhammad R, Farrell M, et al. Relation of morbid obesity and female gender to risk of procedural complications in patients undergoing atrial fibrillation ablation. Am J Cardiol 2013;111:368–73.
- Vinolas X, Freire F, Romero-Menor C, et al. [Predictors of reversion to sinus rhythm previous to electrical cardioversion in patients with persistent atrial fibrillation treated with anti-arrhythmic drugs]. Med Clin (Barc) 2013;140:351–5
- Glover BM, Hong KL, Dagres N, et al. Impact of body mass index on the outcome of catheter ablation of atrial fibrillation. Heart 2019;105:244–50.
- Voskoboinik A, Moskovitch J, Plunkett G, et al. Cardioversion of atrial fibrillation in obese patients: Results from the Cardioversion-BMI randomized controlled trial. J Cardiovasc Electrophysiol 2019;30:155–61.
- Boonyawat K, Caron F, Li A, et al. Association of body weight with efficacy and safety outcomes in phase III randomized controlled trials of direct oral anticoagulants: a systematic review and meta-analysis. J Thromb Haemost 2017;15:1322–33.
- Tittl L, Endig S, Marten S, et al. Impact of BMI on clinical outcomes of NOAC therapy in daily care - Results of the prospective Dresden NOAC Registry (NCT01588119). Int J Cardiol 2018;262:85–91.
- Pathak RK, Middeldorp ME, Meredith M, et al. Long-Term Effect of Goal-Directed Weight Management in an Atrial Fibrillation Cohort: A Long-Term Follow-Up Study (LEGACY). J Am Coll Cardiol 2015; 65:2159–69
- Middeldorp ME, Pathak RK, Meredith M, et al. PREVEntion and regReSsive Effect of weight-loss and risk factor modification on Atrial Fibrillation: the REVERSE-AF study. Eur Eur pacing, arrhythmias, Card Electrophysiol J Work groups Card pacing, arrhythmias, Card Cell Electrophysiol Eur Soc Cardiol 2018;20: 1929–35.
- O’Hare D, O’Neill L, O’Neill MD, et al. Letter to the editor-pREVEntion and regReSsive Effect of weight-loss and risk factor modification on Atrial Fibrillation: the REVERSE-AF study. Eur Eur pacing, arrhythmias, Card Electrophysiol J Work groups Card pacing, arrhythmias, Card Cell Electrophysiol Eur Soc Cardiol 2019; 21:990.
Community Events Calendar


